Five Key Milestones in the History of Research in Cancer Biology
On a cold snowy day in late November 2016, I sat with my father-in-law at the infusion clinic in the local hospital. I was visiting Pennsylvania for my mother-in-law’s funeral. She had recently died of breast cancer, and my father-in-law was receiving treatment for both his prostate cancer and cholangiocarcinoma. Six months later, his battle with cancer ended in his death.
My story, while sad, is unfortunately not unique. Cancer has been around for a long time. Fossil evidence suggests that even dinosaurs had cancer. The ancient Egyptians were the first to record human cancers around 3000 B.C. The name cancer derives from Hippocrates’ observations that the finger-like projections that can appear on tumors look like crab legs. A Roman physician named Celsus translated the Greek term for crab, carcinos, into the Latin word, cancer.
Despite its ubiquity in the history of life on Earth, some equate cancer as a modern disease. As the average life span has increased, the rates of cancer have concomitantly increased, too. With the advent of new technologies and treatments, although the rates of cancer have increased, mortality has generally decreased (although this varies depending on the country).
That brings me to the subject of this blog post – new technologies and treatments. The advancement of science follows technological advances.
In this post, I’m going to talk about five key technological advancements that have advanced research in cancer biology. These technological advances have enhanced our understanding of cancer biology, how we diagnose and treat cancer, or both. I’ll start with the discovery of cells which coincided with the advent of the microscope, and follow major technological advancements up to gene expression profiling with technologies like nCounterⓇ Analysis System and cutting edge spatial biology technologies like the GeoMxⓇ Digital Spatial Profiler and the CosMx™ Spatial Molecular Imager.
Technological Advance #1: The Microscope
Although we usually think of Galileo Galilei and his contributions to the fields of astronomy and physics, he built on the work of a variety of Dutch lens makers to create the first microscope. Later on in the sixteenth century, Anton van Leeuwenhoek (now considered to be the founder of microscopy), was able to create better lenses for microscopes and began to investigate the microscopic world.
Improvements to microscopes continued, and by 1665 lens technology had advanced to the point where one could investigate eukaryotic cells. Robert Hooke is credited with the discovery of the cell-based on his observations of cork samples under a microscope.
Two hundred years later in 1863, Rudolf Virchow came on the scene. Using microscopy, he was able to correctly ascertain for the first time that cancer cells come from normal cells.
Today, microscopy is still a critical technology for cancer diagnosis and for research and development into new treatments. Have you ever had a biopsy before? Or a Pap Smear? A pathologist likely used a microscope to determine if your sample was cancerous or not.
Sophisticated fluorescent microscopy is a cornerstone of modern cancer research. Two cutting-edge spatial biology technologies from NanoString also rely on microscopy – the GeoMx® Digital Spatial Profiler and the CosMx™ Spatial Molecular Imager. But I’ll circle back to these when we get to the fifth key invention, next-generation sequencing.
Technological Advance #2: X-rays and Radiation
In the 1890s two key discoveries were made that radically changed how we treat cancer. 1895 saw Wilhelm Roentgen’s discovery of X-rays and in 1898, Marie and Pierre Curie discovered radium. All three scientists would go on to receive Nobel Prizes for their discoveries.
Doctors realized that radiation was effective at shrinking tumors. In 1900, Thor Stenbeck successfully cured a patient’s skin cancer through small daily doses of radiation. For the first time, patients had a non-surgical option for cancer treatment (surgical treatment made possible through another technological advance, namely general anesthesia).
By the 1940s another treatment would come on the scene – chemotherapy. During World War II, doctors noted that exposure to nitrogen mustard gas would destroy an individual’s white blood cells. In 1946, Alfred Gilman and Louis Goodman noted that nitrogen mustard gas was effective for treating lymphomas and leukemias. In 1949, nitrogen mustard became the first Food and Drug Administration (FDA) approved chemotherapy.
Researchers were able to refine chemotherapies over the following years. In the latter half of the 20th century, scientists were able to develop a variety of targeted therapies for specific cancers. For example, in the late ‘90s, the FDA began approving monoclonal antibodies such as Rituximab and Trastuzumab for non-Hodgkin Lymphoma and breast cancer, respectively.
Technological Advance #3: Sanger Sequencing
Thirty years after Gregor Mendel’s pioneering work in genetics, an ophthalmologist named Hilário de Gouvêa observed that retinoblastoma, a cancer of the eye, was more common in children born to a father with the disease. In 1911, another group noted that breast cancer in mice seemed to run in families.
These experiments seemed to suggest a genetic component to cancer. However, we didn’t know that DNA was the genetic material until the mid-twentieth century, after the classic experiments of Avery, Macleod, and McCarty in 1944, and the Hershey-Chase experiments in 1952. Then in 1953, we saw one of the greatest breakthroughs in the history of molecular biology – the discovery of the structure of DNA. It’s also a story of sexism and scientific misconduct, but that’s the subject of another blog post.
Structure defining function is a major theme of molecular biology. The information carried in DNA is preserved in its structure – the sequence of bases that make up the characteristic ladder shape. In 1977 Frederick Sanger devised a way to figure out the sequence of bases in a DNA molecule for the first time. Then in 1994, over 100 years after de Gouvêa’s observation that cancer appeared to be hereditary, the first gene associated with a cancer, BRCA1, was sequenced. Identifying a genetic basis of cancer was a critical step toward personalized treatments, understanding how cancers can arise, and identifying actionable risk factors for cancer prevention.
Using technologies like NanoString’s nCounter® we can go beyond looking at genes related to cancer risk and instead profile all the transcripts in a single patient’s tumor. This allows for personalized medicine or designing treatment around the individual genetic signature of the tumor. In 2017, the National Cancer Institute kicked off the Pediatric MATCH Precision Medicine Clinical Trial. Pediatric MATCH uses sequencing of solid tumors in children with cancers that are not responding to conventional treatments to design treatments based on the genetic signatures in the tumor, rather than on the cancer’s tissue of origin.
Technological Advance #4: Genetic Engineering
In 1975, doctors began to use chemotherapy alongside radiation and/or surgical therapy creating the so-called “three-legged stool” of cancer treatment that was the standard of care through the end of the 20th century. Now, genetic engineering and immunotherapy are creating a new era of cancer treatment.
Genetic engineering and specifically, immunotherapy, have been involved in cancer research since the 1970s. In 1975, Kohler and Milstein devised a strategy for generating monoclonal antibodies by fusing myeloma and B cells together. These hybridoma cells produced large amounts of antibodies that could be used to treat cancers. Scientists can engineer different hybridomas for treating different kinds of cancers. Since 1985, over 100 monoclonal antibodies gained approval from various governing agencies.
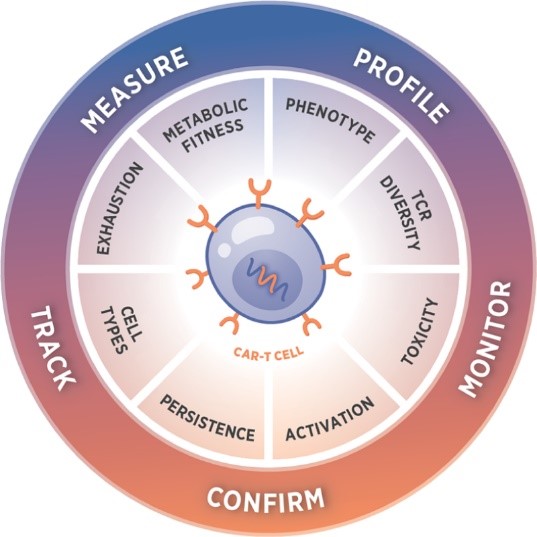
The hottest news today in immunotherapy is chimeric antigen receptor (CAR) T cell therapy. Instead of using genetic engineering to create better treatments, the patient’s own cells are engineered to recognize cancer cells and mount an immune response.
First, doctors collect a patient’s T cells. These T cells are then engineered to have enhanced ability to recognize cancer cells. Then, they are infused back into the patient. Although the technology still can lead to undesirable side effects, the FDA has approved some CAR-T cell protocols.
Technological Advance #5: Spatial Biology
Our last technological leap forward is really the culmination of two advances I’ve already discussed in this article – microscopy and Sanger sequencing. Although Sanger sequencing is still in common use in clinics and laboratories today, it’s limited to a single known sequence. Next-generation sequencing facilitates the sequencing of many genes at once, including unknown genes.
Spatial biology combines cutting-edge microscopy with next-generation sequencing to provide unparalleled insight into tissue structure and cell makeup of tumors. NanoString has developed technologies that allow researchers to examine spatial biology in a variety of contexts. For example, researchers can use the GeoMx® Digital Spatial Profiler to examine the differences in expression in the tumor and the tumor microenvironment. The CosMx™ Spatial Molecular Imager can help researchers to examine a single cell and how it interacts with its neighbors.

Spatial biology provides scientists with a sweet spot for understanding the molecular dynamics of cancer. Depending on the research question of interest, scientists can gain insights quickly into the context of cancer cells, at the level of tissue structures, cell populations, or single cells. It’s exciting to consider how spatial biology will facilitate the next big discovery in cancer research.
The Future of Cancer Research and the Next Technological Advance
From dinosaur bones with tumors to spatial biology, multicellular life on Earth has had a long relationship with cancer. One theme across this blogpost is the importance of genetics, including both genetic engineering and gene sequencing, to our modern understanding of cancer, and how we can treat (and prevent!) cancer.
For years, biologists talked about the $1000 genome – and how cheap genome sequencing will change medicine. Now we’re approaching the $100 genome. Will it be possible for people to regularly have their entire genomes sequenced as part of the standard of care in the near future?
I’m excited for a future when slow, painful, cancer deaths are a thing of the past when the story of my in-laws serves not as a tale that many can relate to, but as a reminder of how far we’ve come.
GeoMx® Digital Spatial Profiler and CosMx™ Spatial Molecular Imager are for Research Use Only. Not for use in diagnostic procedures.