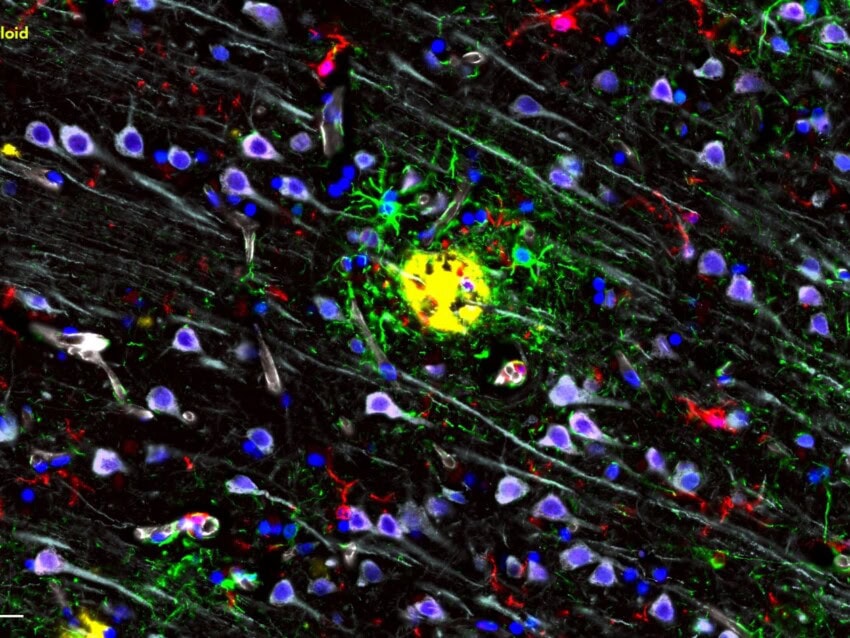
Q&A with Dr. Stefan Prokop – Alzheimer’s: From the Amyloid Hypothesis to the Cellular Phase and Beyond

Stefan Prokop, MD, is a Neuropathologist and Research Fellow at the University of Pennsylvania School of Medicine. His work is focused on Alzheimer’s Disease (AD) with a specific interest in the role of the immune system in AD pathogenesis and progression. We recently spoke with Dr. Prokop about his research and the state of the field.
NanoString (NS): Can you share your research history that led you to this current project focused on AD and microglia?
Stefan Prokop (SP): I have been working in the AD field for more than 15 years and have focused for the last 10 years on the role of the innate immune system in AD pathogenesis and progression. Most of my previous work was centered around mouse models with AD-like pathology. I was involved in early studies testing the impact of microglia depletion on Abeta plaque-pathology and used this model to explore the potential usability of peripheral myeloid cells to combat Abeta deposits. As a neuropathologist I am confronted daily with the complexity of neurodegenerative diseases in human brains and the apparent deficiency of commonly used mouse models to mimic the human condition, particularly with respect to microglia function. To bridge the gap between microglia studies in mouse models and the human disease condition, we set out to profile microglia across the stages of AD progression to set a baseline for differences and similarities in the innate immune response in AD between humans and mice.
Beyond the scope of my present project, I aim to establish and apply novel methods to interrogate human tissue specimens with respect to immune activation and the interplay between different cell types in aging and disease.
NS: Microglia have taken center stage in neurodegenerative disease research in recent years. What is your perspective on classical hypotheses of AD pathogenesis and how does the recent microglia focus fit in?
SP: I am a firm believer in the hypothesis that aggregation of proteins is at the center of pathogenesis of common neurodegenerative diseases. The recent microglia hype has demonstrated that we are still far from understanding how complex interactions of all the cell types in the brain, including microglia and astrocytes, modulate the toxicity of aggregated proteins in the human brain and thus ultimately dictate an individual’s age of onset, disease progression, and clinical phenotype.
NS: What is your opinion on the ability to translate findings about microglia biology in neurodegenerative disease from mouse models to the human condition?
SP: Work in mouse models has elevated our knowledge of microglia biology, in particular with respect to the pathogenesis and progression of AD, and the next crucial step for translation is the validation of these findings in human subjects, biofluids, and tissue specimens to move the most promising pathways forward. I am convinced that the immune system will be a powerful ally in combatting neurodegenerative diseases, as we have seen the tremendous success of immune modulators in cancer therapy, and we may be closer to reaching a breakthrough than we think.
NS: Your work focuses on the microglia response to various types of protein aggregates. Do you anticipate that the microglia response to these varied proteins will be different across diseases, or do you foresee a common therapy that may address microglia function/dysfunction in many common neurodegenerative diseases?
SP: One of the central aspects of my research is to compare/contrast the innate immune response across the spectrum of neurodegenerative diseases, and our preliminary data show that there are profound differences in the immune response across neurodegenerative diseases, but also within one specific disease. I believe that the interplay between the pathologic protein and underlying genetic and other medical conditions of the individual determines the response of microglia. We will have to identify these patterns of response to enable individualized therapeutic interventions that may target the same pathway for different underlying protein pathologies but different pathways for patient subtypes within one disease entity.
NS: What do you feel is the biggest challenge in current neurodegenerative disease research?
SP: How to translate the findings of basic AD research into preventative or disease-modifying therapies for neurodegenerative diseases.
NS: Why did you decide to start using NanoString® in your work?
SP: We were looking at the brain region-specific immune activation profile of sorted microglia in contrast to whole tissue samples from a mouse model of alpha-Synuclein aggregation. NanoString provided a fast and robust platform to interrogate a multitude of immune-related genes and pathways in our limited sample.
NS: What are the biggest benefits of employing NanoString analysis in your work?
SP: For the gene expression analysis, the ability to get reproducible and robust results from FFPE tissue allows for a) direct comparison of conventional histology with gene expression patterns in the same sample and b) availability of more tissue samples for analysis, especially archival FFPE tissue of rare genetic variants. The robustness of the NanoString platform, the fact that it allows for the use of FFPE tissue and fragmented DNA or RNA targets, and has minimal tissue input requirements, combined with the speed of results generation are all key advantages over current sequencing platforms.
Additionally, multiplexed quantitative protein analysis is one of the biggest needs in neuroimmunology research. The GeoMx™ Digital Spatial Profiler (DSP) not only allows for the quantification of more than 35 proteins in one sample but also enables spatially resolved mapping of the quantified proteins. Furthermore, it is applicable to FFPE tissue samples too, which enables the inclusion of more cases than with other assays relying on frozen tissue and facilitates the direct comparison of multiplexed protein quantification with conventional histology and NanoString gene expression assays.
NS: What is the most surprising findings so far using GeoMx DSP?
SP: The data are very preliminary at this stage; however, I was pleased to see that our biochemical and histological data nicely correlate with the GeoMx DSP data, which gives confidence in the results. Beyond that, I’ve also begun to explore differences in protein aggregation in Trem2 variants that seem to have been picked up in DSP, but that we weren’t able to analyze with our conventional methods. I’m looking forward to digging into this data further!
NS: What’s next for this work?
SP: The next step is to apply this combined multimodal analysis of tissue specimens that we’ve now established to larger cohorts of AD patients to identify subtypes of sporadic AD, and to apply these analytical methods to samples of patients suffering from related dementias to compare/contrast the cellular response in different neurodegenerative diseases.
NS: What is your take on the current state of the field? Where are we now, and where do we need to go?
SP: We’ve come a long way from the initial description of pathologic findings in the brain of the first AD patient more than 100 years ago, to the identification of the aggregated proteins (Abeta and Tau) underlying the pathologic findings, to the identification of numerous genetic risk factors and modifiers, finally leading to the recognition of a “cellular phase” of AD progression centrally involving immune cells. The next step will be to combine all these findings and understand the interplay between the aggregated proteins and individual modifiers, including genetic variants and concurrent diseases, to identify subtypes of AD driven by different underlying cellular mechanisms. Furthermore, it will be important to study early disease stages and better understand initiating events of protein aggregation many decades before actual symptoms appear to know when and how to intervene to prevent disease onset.
To learn more about Dr. Stefan Prokop’s work, view the webinar “Comprehensive analysis of microglia across neuropathological stages of Alzheimer’s disease (AD)”
FOR RESEARCH USE ONLY. Not for use in diagnostic procedures.