Integrated Spatial Multiomics Deconvolve Multicellular Dynamics in Pancreatic Cancer
In an extraordinary team effort led by the Center for Systems Biology at Harvard Medical School, in collaboration with NanoString, scientist Will Hwang and colleagues used integrated spatial multiomic and spatial analysis to understand pancreatic ductal adenocarcinoma (PDAC): with single nucleus RNA seq, the spatial multiomics platform GeoMx® Digital Spatial Profiler with the Whole Transcriptome Atlas (WTA) they profiled tumor samples from a cohort of pancreatic cancer patients before or after neoadjuvant therapy.
We already described some of the key characteristics of PDAC and why its high mortality rate is also the reason why it is so hard to study. We also introduced a groundbreaking paper published by Harvard scientists where they revealed many potential therapeutic targets. In part two of this blog miniseries, we will look in more detail at the disruptive work Dr. Hwang and colleagues published in Nature Genetic on July 28, 2022.
Last week’s blog taught us that pancreatic cancer is anatomically hard to reach and study even with the most current technologies. Previous studies have lacked the depth, multiplexing ability, and spatial resolution needed to reveal the diversity of both malignant and non-malignant cells within the tumor. The team behind this Nature Genetics paper did just that by integrating single nuclei RNA sequencing results with that from the GeoMx® Digital Spatial Profiler (DSP).
The study population consisted of forty-three banked frozen, histologically confirmed, primary PDAC specimens from 43 patients, 18 untreated and 25 treated. The 25 treated patients included two groups divided based on treatment: 14 received standard chemotherapy followed by radiotherapy (called the CRT cohort), and 11 patients were enrolled in a clinical trial evaluating the effectiveness of a neoadjuvant (Losartan) added to the standard therapy (CRTL).
Exploring the Transcriptome to Uncover Treatment-Associated Changes
Initial unsupervised clustering of single-nuclei transcriptomes identified 33 cell subsets annotated by gene signatures comprised of malignant and non-malignant cells. Cell types included immune cells, CAFs (i.e., the cancer-associated fibroblasts that deposit extracellular matrix in the microenvironment), endothelial cells, and vascular smooth muscle cells. The team also found a small subset of acinar cells expressing high levels of genes that promote pancreatic inflammation, ADM – the acinar cells on their way to becoming neoplastic – and pancreatic intraepithelial neoplasia (PanIN).
Within the treated group, samples from patients receiving neo-adjuvant therapy in combination with Losartan (CRTL) expressed an increased effector CD8 phenotype with effector functions and lower expression of regulatory genes, an observation consistent with the effect of Losartan that increases cytotoxic activity.
ADM and ductal cells showed a chronic inflammatory phenotype, with higher expression of genes upregulated by KRAS pathway activation, a signature typical in ADM and atypical ductal cells in the intermediate stages of PDAC tumorigenesis.
These findings support the known path of transformation from acinar to ADM to ductal to atypical ductal to malignant cells.
Expanding and Redefining Malignant Cell Taxonomy May Lead to New Therapeutic Targets.
snRNASeq can quantify transcripts in individual cells, but each cell’s profile includes both cell-type specific programs and broader cellular activities such as life cycle or response to environmental processes. The two are hard to disentangle without the use of specific algorithms.
The consensus non-negative matrix factorization (cNMF) does exactly that: it infers the identity and activity of cellular signaling programs and their relative contributions in each cell. The authors used it to identify multiple gene expression programs exhibited by malignant cells and fibroblasts: 14 malignant programs and 4 CAFs programs, thus redefining and expanding the cell taxonomy of malignant cells and fibroblasts in pancreatic cancer.
The authors discovered a post-treatment program in both CRT and CRTL patient cohorts, the neural-like progenitor (NRP) malignant program enriched with residual tumor cells. These NRP program cells are resistant to treatment, making them a program associated with failure to eradicate the tumor completely.
Moreover, when they assessed the prognostic relevance of the malignant and CAFs programs, NRP was associated with a shorter time to progression (TTP) – the time from randomization to tumor progression –and overall survival. In contrast, immunomodulatory programs were associated with longer TTP. This might be a future therapeutic target.
Neoadjuvant therapy was also associated with differential expression of malignant and CAFs programs, another indication that some cells have a program that makes them resistant to treatment.
From Transcriptome to Spatial Transcriptome: Building a Landscape of Spatial Communities
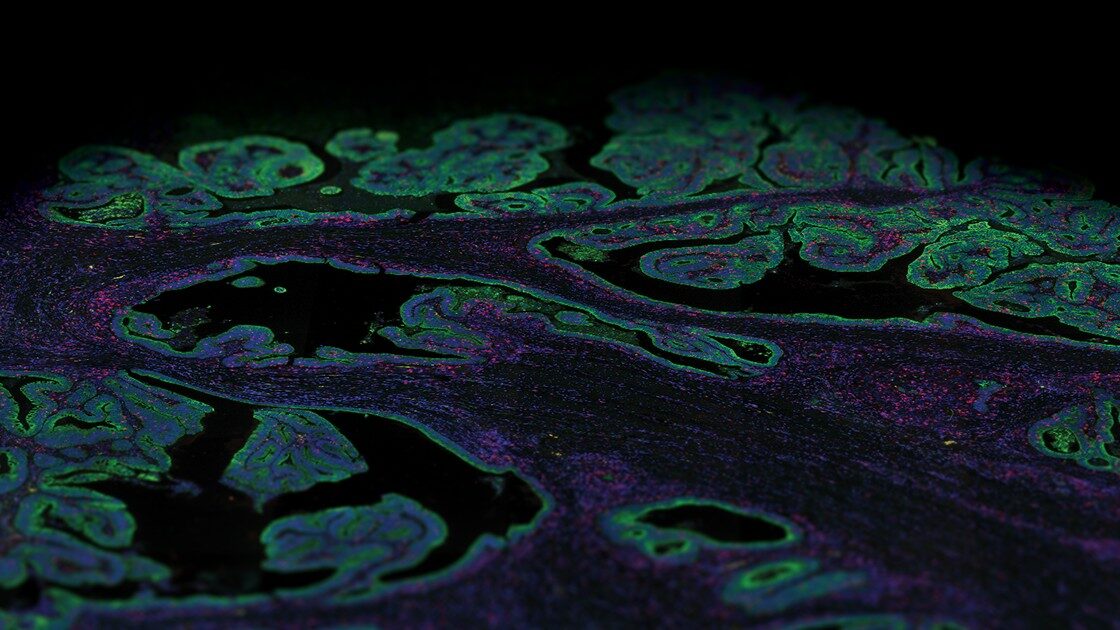
The next question the researchers asked was: are there multicellular spatial associations among these cells? To answer the question, spatial transcriptomics using the GeoMx Digital Spatial Profiler and Whole Transcriptome Atlas (WTA) was used in 21 independent PDAC FFPE specimens, 18 of them with matching snRNA-seq data: 7 treated and 14 untreated.
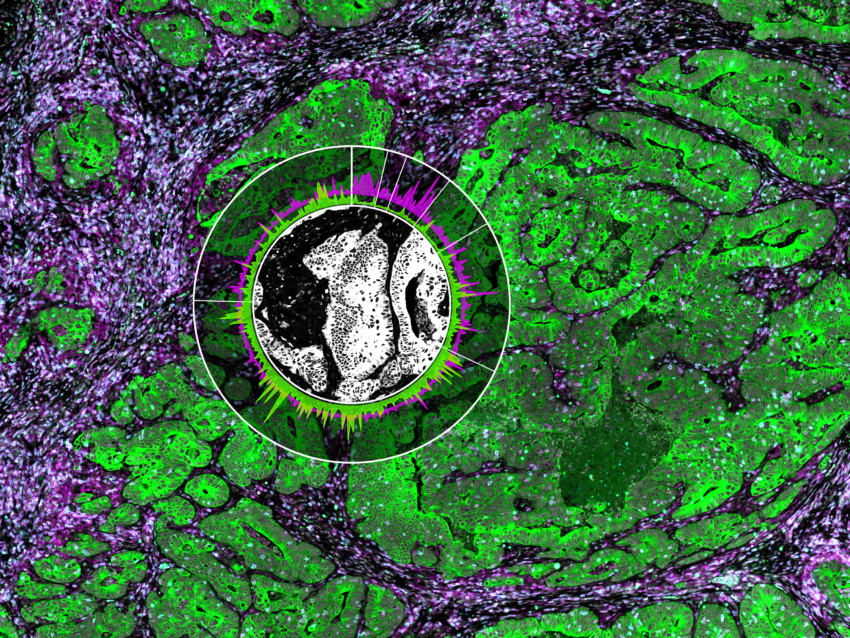
The tissues were labeled with specific morphology markers to define regions of interest (ROIs) within the FFPE samples. These are fluorescently-labeled antibody probes designed by NanoString to identify biological structures and guide the selection and segmentation of distinct biological compartments for whole transcriptome spatial analysis. In this case, the selection strategy involved four morphology markers delineating nuclear, epithelial, immune, and fibroblast compartments.
Spatial Transcriptomics and the Deconvolution of the PDAC Microenvironment
The main components of the PDAC microenvironment are cancer-associated-fibroblasts (CAFs) that create desmoplastic stroma, various immune cells (effector and regulatory T cells, dendritic cells, macrophages, monocytes), and malignant cells.
To identify how the different malignant and stromal programs relate to the composition of the immune cells and the percent ROI area they occupied, the authors correlated them to each other. With GeoMx DSP and the power of spatial transcriptomics, they identified three communities of cells with different contributions from malignant cells, fibroblasts, and immune cells. Community One was a treatment-enriched community characterized by colocalization of the NRP malignant program, neurotropic CAF program, and CD8+ T cells. This suggests an interplay among these cell types/states and confirms that the NRP malignant program, the CAFs, and the cytotoxic T cells are indeed colocalized. This provided an essential spatial context to the discoveries made with RNA sequencing.
In Community Two – basaloid-squamoid – squamoid and basaloid malignant cell programs colocalized with myeloid, lymphoid, and epithelial programs, whereas Community Three – classical – was enriched in classic malignant programs, with myelofibroblastic progenitors and CAFs programs, neutrophils, and dendritic cells, with a low number of immune cells compared to the other communities.
These results are crucial as they reveal for the first time that malignant pancreatic cells follow different cellular programs and cluster in different immune niches within the same tumor.
Going into the Details of Cell Crosstalk
Knowing the spatial associations between fibroblasts, malignant cells, and immune cells gives us the chance to understand how cells communicate with each other. Ligand-receptor interactions among cells can uncover relationships conducive to therapeutic response or resistance: knowing them provides potential targets for better therapeutic interventions.
Several spatially defined receptor-ligand bonds were found differentially expressed across ROIs and were enriched according to treatment status, offering a potential explanation for therapeutic resistance and potential targets for intervention. One to note, CXCL12 and CXCR4 differentially correlated in untreated tumors compared to the treated.
This is important because it suggests a possibility for AMD3100, a known CXCR4 inhibitor, to be used in addition to standard adjuvant therapy.
A New Era for PDAC Targeted Therapies.
Therapies can be more effective if they target specific phenotypes. This consequential paper is redefining the way pancreatic cancer is viewed and provides the new actionable therapeutic targets that are most needed.
A previously unknown malignant program, the NRP program that appears only after treatment in residual cells provides scientists with a new target for better adjuvant therapies.
This paper described the discovery of niches within the cancer tissue that host different malignant programs with different immune cells – some malignant programs are close to CD8 T cells, myeloid or dendritic cells – indicating immune infiltration occurs with different immune cells, offering different strategies to get past immune suppression.
Knowing how ligand-receptor pairs interact in space can help target specific receptors as an addition to adjuvant therapy to ultimately treat, if not cure, one of the most devastating cancers.
Contact us if you want to know more about spatial transcriptomics, GeoMx DSP, and all its applications, including the use of the WTA in this seminal work, or you can listen directly to Dr. Hwang!
And if you want to discover more about tissues and keep track of your spatial experiments throughout 2023, order the NanoString calendar and discover the winners of our annual spatial biology image contest, complete with the scientific stories behind every image, one month at a time!
For Research Use Only. Not for use in diagnostic procedures.