COVID-19 Pathogenesis Steps Out of the Shadows
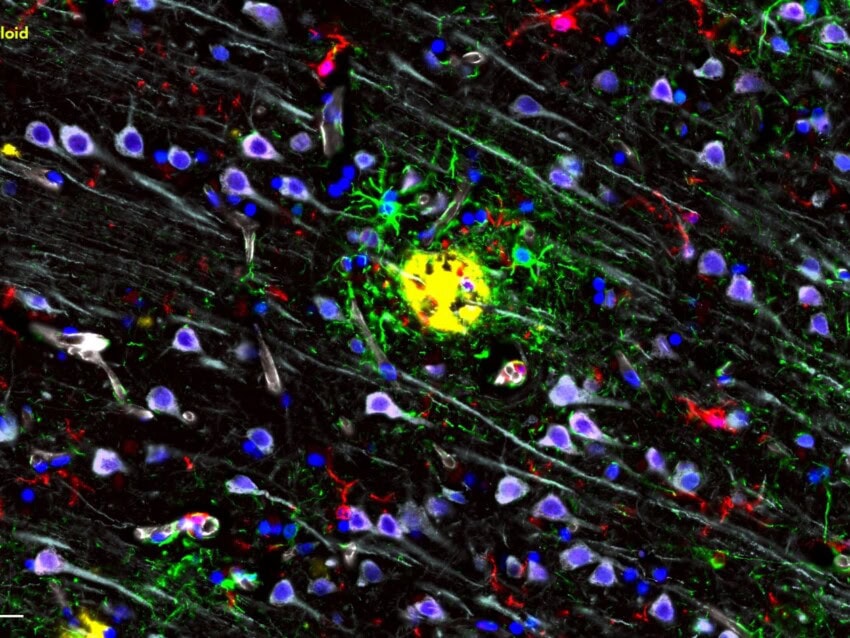
“Analysis of pulmonary vessels showed that widespread thrombosis with microangiopathy ─ a severe condition wherein small blood clots are formed within the tiniest of blood vessels ─ was 9x more prevalent in COVID-19 patients”
Do you even remember “life before COVID-19”? Many of us have come to terms with what unfortunately is now the new normal: social distancing, wearing face masks, and working from home.
As the daily counts of new infections and casualties begin to sound like reports from the battlefield, scientists around the world are working around the clock to study the SARS-CoV-2 virus: collect patient samples, run experiments, and analyze the data to understand the virus’ behavior in cells and its relationship with the immune system; this information is critical to developing a vaccine and finding a cure.
When we come across publications from our customers where the dynamics between the coronavirus and the host immune response is profiled with nCounter® gene expression panels, the initial reaction of many of us here at NanoString is pride and joy in that we are helping our customers be a part of the solution to this global pandemic.
This blog post highlights some of these papers: we want to recognize and thank these scientists who trusted NanoString with precious and scarce COVID-19 samples in this extraordinary situation when saving time and maximizing resources is critical.
Type I IFN immunoprofiling in COVID-19 patients (1)
After a pilot study revealed that IL-6, CRP and IFN-g-Induced protein 10 (IP-10) were increased in 2 patients with symptomatic disease, while no IFN- α2 was detected when compared to a patient with mild symptoms, the research team led by Sophie Trouillet-Assant, Ph.D. profiled the immune response of 26 critically ill ICU patients.
Time-course measurements of plasma cytokines showed a peak in IFN-α2 at day 8-10 after the onset of symptoms that corresponded to the viral replication phase and decreased over time. Conversely, a subset of patients (n=5, 19%) showed sustained abrogation of IFN-I production.
Furthermore, IFN-α2 positively correlated with interferon-stimulated genes (ISGs). An IFN signature score defined by six ISGs was included in a Custom CodeSet and obtained from four COVID-19 patients.
There was a strong pro-inflammatory response in all cases (CRP, IL-6 or IP-10), which started early and remained positive, while the type I IFN response decreased after 10 days of infection. The five patients with no IFN-α production presented poorer outcomes, requiring invasive ventilation and a longer ICU stay. Viral load tended to be higher in IFN-negative COVID-19 patients at the time of diagnosis.
These data show a heterogeneous pattern of IFN-α response in COVID-19 patients. The IFN-I response was impaired in about one-fifth of critically ill patients, which may be associated with poorer outcomes.
Pulmonary Vascular Endothelialitis, Thrombosis, and Angiogenesis in Covid-19 (2)
In this seminal study published in the New England Journal of Medicine, the authors demonstrated that vascular angiogenesis distinguishes the pulmonary pathobiology of COVID-19 from that of equally severe influenza virus infection.
The authors explored the morphologic and molecular changes in the lung tissue of seven patients who died from COVID-19 and compared them to the lungs of seven patients who died from acute respiratory distress syndrome (ARDS) secondary to influenza A (H1N1) infection and to 10 age-matched, uninfected control lungs. Lung tissue sections were studied with seven-color immunohistochemistry, micro-computed tomographic imaging, scanning electron microscopy, corrosion casting, and direct multiplexed measurement of gene expression using two nCounter panels.
The patients who died from COVID-19–associated or influenza-associated respiratory failure revealed perivascular T cell infiltration and alveolar damage, but the lungs from COVID-19 patients also exhibited severe endothelial injury associated with the presence of intracellular virus and a higher number of CD4+ T cells compared to lungs from patients with influenza.
All COVID-19 lung tissues had diffuse alveolar damage with necrosis of the alveolar lining, along with pneumocytes hyperplasia and linear intra-alveolar fibrin deposition. Differential gene expression analysis of 249 genes with the nCounter Inflammation Panel revealed that while several genes were differentially expressed in both COVID-19 and influenza lung tissue samples, 79 inflammation-related genes were differentially regulated only in COVID-19 specimens.
Analysis of pulmonary vessels showed that widespread thrombosis with microangiopathy ─ a severe condition wherein small blood clots are formed within the tiniest of blood vessels ─ was 9x more prevalent in COVID-19 patients, and new vessels were growing through a mechanism called intussusceptive angiogenesis — a term that refers to forming new blood vessels by splitting an existing blood vessel in two. Multiplexed analysis of 323 angiogenesis-related genes from the nCounter PanCancer Progression Panel revealed 69 angiogenesis-related genes were differentially regulated in the COVID-19 tissue samples.
While this study may be limited by the small number of samples, the results are a remarkable step toward gaining knowledge that may improve the clinical outcome of patients.
Impaired type I interferon activity and inflammatory responses in severe COVID-19 patients (3)
In this paper published on Science and cited by Conor Gruber in Nature Reviews Immunology, Jérôme Hadjadj and colleagues performed an integrated immune analysis on whole blood which included profiling for immune cell phenotypes, apoptosis, and cytokines on a cohort of 50 COVID-19 patients with a spectrum of disease severity. The cohort was controlled for age, duration of symptoms, and comorbidities, and the samples were tested in the absence of anti-inflammatory therapies.
Flow cytometry revealed subsets of patients with decreased T cells as well as increased CD38+ and HLA-DR+ activity in activated T cells from infected patients; a modest PD-1 increase associated with disease severity. Annexin-V expression as seen by flow cytometry and upregulation of apoptosis genes supported the hypothesis that lymphocytopenia may be partly explained by an increase in T cell apoptosis.
Expression analysis of 574 genes included in the nCounter Human Immunology Panel identified differentially expressed genes as a function of disease severity: the first principal component (PC1) was driven by inflammatory and innate immune response genes and distinguished patients with severe disease from the remainder of patients, while PC2 was enriched in genes encoding proteins involved in both type I and type II interferon (IFN) responses and distinguished mild-to-moderate patients, suggesting a severity grade-dependent increase in the activation of innate and inflammatory pathways.
Critically ill patients showed a profoundly impaired type I IFN response characterized by low interferon production and activity with a consequent downregulation of IFN-stimulated genes after IFN-α stimulation. Low interferon was associated with persistent blood virus load, an exacerbated inflammatory response partially driven by NFκB, TNF-α, IL-6 production and signaling, and high inflammatory chemokines.
A dynamic immune response shapes COVID-19 progression (4)
In this study led by Eugenia Ziying Ong, the authors saw an important role for T cells in COVID-19 pathogenesis.
Three COVID-19 patients with different degrees of disease severity were analyzed daily for the presence of the virus and gene expression changes. Ten healthy volunteers were included as controls. Throat swabs and blood draws were taken daily from the patients, followed by RNA extraction and storage at -80°C. Viral presence was quantified by RT-PCR. Gene expression was measured with the nCounter Human Immunology Panel and analyzed with nSolver™ Data Analysis Software version 4.0.
One patient, a 67-year-old male, was severely ill and had decreased lung function. The other two cases were mild, and only one was febrile. Interestingly, the duration of the infection was negatively correlated with the severity of the disease.
Transcriptional changes in the three COVID-19 patients and the ten healthy volunteers revealed a dynamic expression of pro-inflammatory genes: for the severely ill patient, expression of most of these genes peaked after the nadir of respiratory function, while only expression of the cytokines IL1A and IL1B preceded the nadir of respiratory function; furthermore, expression of the IL1 receptor and associated downstream signaling molecules were also induced before the respiratory function nadir, raising the possibility that the IL1 pathway may be a suitable set of biomarkers for severe respiratory disease.
Another remarkable observation was a decrease in CD4, CD8A, and CD8B transcripts. Also, MHC Class II and T cell activation pathways were decreased in the critically ill patient compared to the milder cases and healthy controls.
These data show an association between reduced activation of adaptive immune genes and an exacerbated pro-inflammatory response, whereas the attenuated pro-inflammatory response observed in the milder cases not only may have elicited mild symptoms but also may have led to attenuated T cell activation, coinciding with prolonged viral positivity.
A shifting paradigm
The understanding of COVID-19 is still in its infancy despite recent scientific breakthroughs, and new observations are now challenging the notion that dampening the inflammatory response is the best course of action in all patients.
While anti-inflammatory and IFN-based therapies are the leading treatment options for the critically ill, some doctors treating cancer patients with COVID-19 are observing an unexpected phenomenon: T cell exhaustion (5). In fact, as T cells battle the infection, they become metabolically exhausted and dysfunctional; in normal conditions, they would be replaced, but with severe COVID-19 they never do, allowing uncontrolled viral replication. This line of thinking is leading to a more nuanced approach to immune modulation that addresses the dynamics of the host response as well as viral replication so that the initial anti-inflammatory response is tamped down and the adaptive immune response is bolstered.
Study the dynamics of the host response to SARS-CoV-2 with the NEW Human nCounter Host Response Panel – profile 785 genes across 50+ pathways involved in the five major components of the immune response to infectious disease with pathogen-agnostic content optimized for blood but suitable for all sample types. Pair the Host Response Panel with the 20-gene Coronavirus Panel Plus to additionally detect coronavirus transcripts and expression of the Human ACE2 receptor or create your own Panel Plus spike-in of up to 30 genes by mixing and matching content of your choice to fit your research project; for example, add on genes for different organ systems to study COVID-19 tissue damage.
You can read the complete papers here:
- Trouillet-Assant et al., Type I IFN immunoprofiling in COVID-19 patients. 2020 Apr 29, J Allergy Clin Immunol.
- Maximilian Ackermann et al., Pulmonary Vascular Endothelialitis, Thrombosis, and Angiogenesis in Covid-19. May 21, 2020, NEJM.org. DOI: 10.1056/NEJMoa2015432
- Jérôme Hadjadj et al., Impaired type I interferon activity and inflammatory responses in severe COVID-19 patients. Science 13 Jul 2020: eabc6027 DOI: 10.1126/science.abc6027
- Ong et al., A Dynamic Immune Response Shapes COVID-19 Progression. Cell Host & Microbe27, 1–4 June 10, 2020
- 5) Megan Cully, Immune status could determine efficacy of COVID-19 therapies. Nature Reviews Drug Discovery. doi: 10.1038/d41573-020-00110-3
FOR RESEARCH USE ONLY. Not for use in diagnostic procedures